 without the addition of HCl, is supplied to the cell. The value of Ce, which is referred to as the current efficiency, thus becomes a determining factor in the economics of the ion exchange membrane process. The membrane effectively allows the passage of Na+-ions only and prevents the diffusion of anions from the anode compartment to the cathode compartment, thereby making it possible to obtain caustic soda of very high purity.ġ Faraday of electricity produces 1 equivalent of Cl2 (theoretically, when no Oxygen is discharged on the anode, and when feed brine is acidified) in the anode compartment and 1 equivalent of H 2 and Ce equivalent of NaOH in the cathode compartment. It is selectively permeable to Na+-Cations and the passage of OH – ions back into the anode compartment is blocked, thus allowing a high current efficiency. The ion exchange membrane is impermeable for liquid and gas. 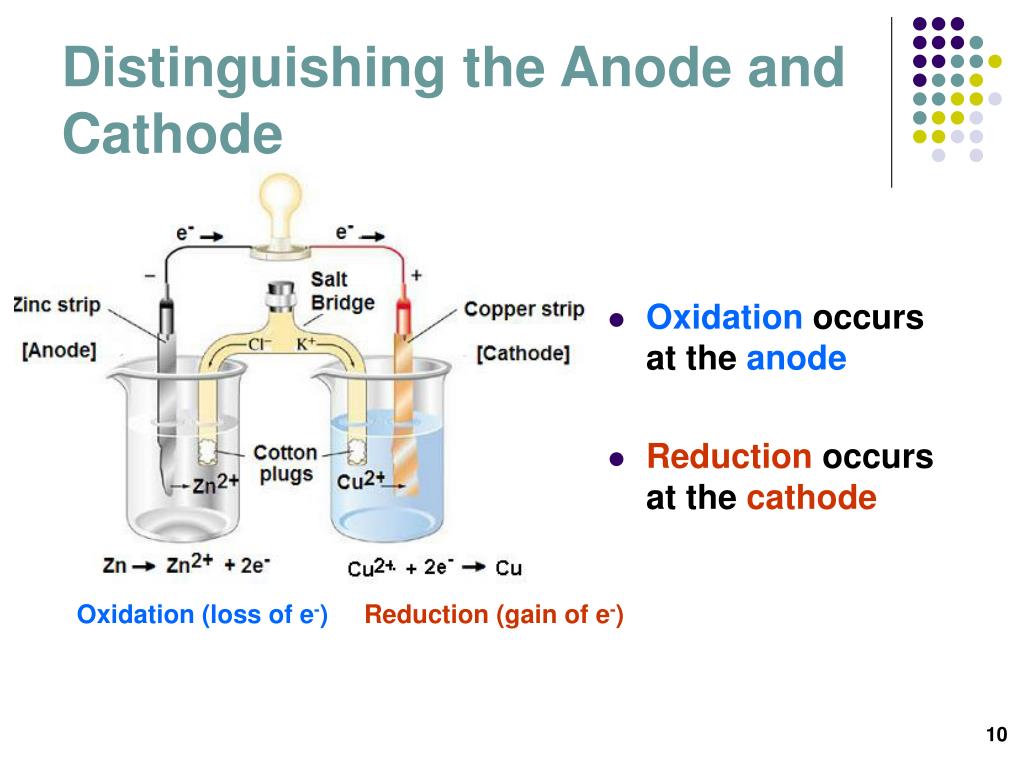
The physical and electrochemical properties and ion-exchange capability can vary widely. Membranes are currently available to produce caustic concentrations of up to 33% NaOH with optimum performance. The chemical composition of the membrane, based on fluorocarbon matrices, defines the range of caustic concentration at which optimum operating performance is given. The ion exchange membrane is the heart of the membrane cell system. It acts as a partition between the anode and the cathode compartment of the cell. The effectiveness of the membrane as a separation device between the anode and the cathode compartments defines the current efficiency of the cell. Principles of the Membrane Process for chlor alkali process Rack :- CS Structure housing 34 elements.Įlement :- Assembly of one each of Cathode, membrane and anode. Definitions related to chloralkali PlantĬhloralkali Plant is a Chemical Plant comprising all equipment and Chloralkali process for the production and Treatment of Chlorine, hydrogen, and Caustic Soda ( Sodium hydroxide).Ĭell Room:- Unit comprising electrolyzes.Įlectrolysers (cell) :- Membrane Cell Package. The direction of electron flow in the external circuit is always from the anode (+) to Cathode (-). , Cl- –> ½ Cl2 + e – ) is the anode the electrode at which an electron consuming reaction occurs (e.g., H 2O + e- –> ½ H2 + OH – ) is called the cathode. The electrode at which an electron-producing ionic reaction occurs (e.g. The electric circuit through the cell is completed by electrolytes that support charge transfer by ionic conduction. In operation, a cell (or a series of cells) is connected to an external voltage source (rectifier unit), and the charge is transferred by electrons between electrodes through the external circuit. These phases are called electrodes and must be good electricity conductors.
0 Comments
Leave a Reply. |


 RSS Feed
RSS Feed
